| Structure | Name/CAS No. | Articles |
|---|---|---|
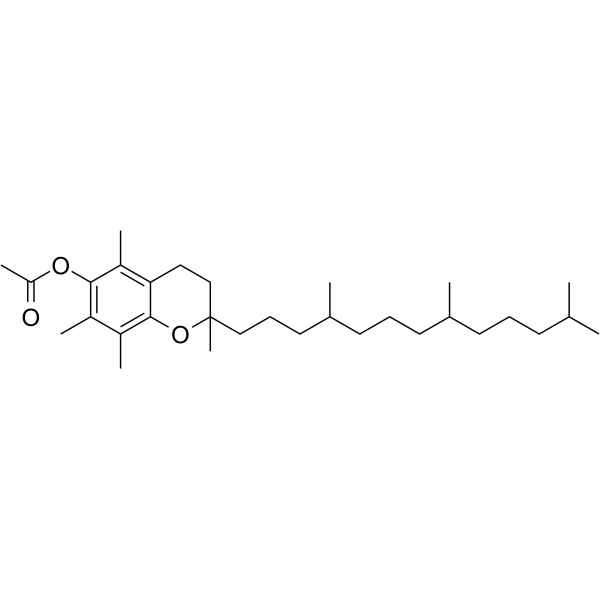 |
Tocopheryl acetate
CAS:7695-91-2 |
|
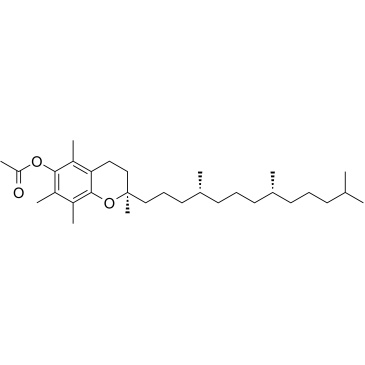 |
alpha-Tocopherol acetate
CAS:58-95-7 |