| Structure | Name/CAS No. | Articles |
|---|---|---|
 |
Methyl paraoxon
CAS:950-35-6 |
|
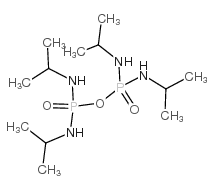 |
N,N',N'',N'''-tetraisopropyldiphosphoramide
CAS:513-00-8 |