Semisynthesis and antifeedant activity of new derivatives of a dihydro-β-agarofuran from Parnassia wightiana.
Jiang-Jiang Tang, Fei-Yu Zhang, Dong-Mei Wang, Jun-Mian Tian, Shuai Dong, Jin-Ming Gao
Index: Int. J. Mol. Sci. 14 , 19484-93, (2013)
Full Text: HTML
Abstract
Five new derivatives (2-6) were semi-synthesized using compound 1, a dihydro-β-agarofuran sesquiterpene with C-2 ketone obtained from Parnassia wightiana, as the starting material by acylation, oxidation, reduction, esterification, and amination, respectively. Structures of 2-6 were confirmed by 1D- and 2D-NMR and HR-ESI-MS spectra. In addition, antifeedant activities of these compounds (1-6) were tested against the 3rd-instar larvae of Mythimna separata. Antifeedant effects of compounds 2 and 4 were greater than the parent compound 1 whereas other compounds exhibited low to no feeding deterrent effects against third instar M. separata larvae in lab bioassays. Therefore, our results suggest that acylated and reduced derivatives at C-8 and C-2, respectively, of 1 may improve the antifeeding effect. This preliminary information will be useful in designing new insect control agents against M. separata and other important pests.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
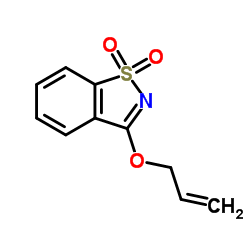 |
3-(Allyloxy)-1,2-benzothiazole 1,1-dioxide
CAS:27605-76-1 |
C10H9NO3S |
|
Characterization of PBZ1, a probenazole-inducible gene, in s...
2001-01-01 [Biosci. Biotechnol. Biochem. 65(1) , 205-8, (2001)] |
|
Different responses of two genes associated with disease res...
2009-01-01 [Curr. Issues Mol. Biol. 11 Suppl 1 , i85-94, (2009)] |
|
Proteome analysis of probenazole-effect in rice-bacterial bl...
2009-01-01 [Protein Pept. Lett. 16(9) , 1041-52, (2009)] |
|
Occupational contact dermatitis caused by probenazole in agr...
2009-12-01 [Contact Dermatitis 61(6) , 350-1, (2009)] |
|
A novel rice PR10 protein, RSOsPR10, specifically induced in...
2004-05-01 [Plant Cell Physiol. 45(5) , 550-9, (2004)] |