FeCl3 mediated arylidenation of carbohydrates.
Nabamita Basu, Sajal K Maity, Soumik Roy, Shuvendu Singha, Rina Ghosh
Index: Carbohydr. Res. 346(5) , 534-9, (2011)
Full Text: HTML
Abstract
Glycosides and thioglycosides based on monosaccharides in reaction with benzaldehyde dimethylacetal or p-methoxybenzaldehyde dimethyl acetal undergo FeCl(3)-catalyzed (20 mol%) regioselective 4,6-O-arylidenation producing the corresponding acetals in high yields. FeCl(3) also mediates acetalation of glycosides and thioglycosides of cellobiose, maltose, and lactose affording the corresponding 4',6'-O-benzylidene acetals, which were isolated after their acetylation in situ with acetic anhydride and pyridine. The combined yields (two steps) of these final products are also high (61-84%). The procedure is applicable to a wide variety of functional groups including -OBn.Copyright © 2011 Elsevier Ltd. All rights reserved.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
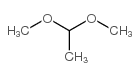 |
Dimethyl Acetal
CAS:534-15-6 |
C4H10O2 |
|
One-pot enol silane formation-Mukaiyama aldol-type addition ...
2008-04-18 [J. Org. Chem. 73(8) , 3299-302, (2008)] |
|
An expeditious synthesis of 4-hydroxy-2E-nonenal (4-HNE), it...
2007-12-01 [Chem. Phys. Lipids 150(2) , 239-43, (2007)] |
|
Matrix isolation infrared and ab initio study of the conform...
2003-05-01 [Spectrochim. Acta. A. Mol. Biomol. Spectrosc. 59(7) , 1497-507, (2003)] |
|
Hybrid Lithium-Sulfur Batteries with a Solid Electrolyte Mem...
2015-08-05 [ACS Appl. Mater. Interfaces 7 , 16625-31, (2015)] |
|
Mice lacking mitochondrial ferritin are more sensitive to do...
2014-08-01 [J. Mol. Med. 92(8) , 859-69, (2014)] |