A synthesis of 4-hydroxy-2-trans-nonenal and 4-(3H) 4-hydroxy-2-trans-nonenal.
A Chandra, S K Srivastava
Index: Lipids 32(7) , 779-82, (1997)
Full Text: HTML
Abstract
4-Hydroxy-2-trans-nonenal, the most abundant and toxic unsaturated aldehyde generated during membrane lipid peroxidation, was synthesized starting from fumaraldehyde dimethyl acetal. In the first step of the synthesis, the fumaraldehyde dimethyl acetal was partially hydrolyzed using amberlyst catalyst to obtain the monoacetal. The 4-hydroxy-2-trans-nonenal was synthesized by the Grignard reaction of the fumaraldehyde monoacetal with 1-bromopentane. 4-Hydroxy-2-trans-nonenal, obtained as its dimethylacetal, was oxidized to its corresponding 4-keto derivative using pyridinium chlorochromate buffered with sodium acetate as the oxidizing agent. 4-(3H) 4-Hydroxy-2-trans-nonenal was obtained in one step by the sodium borotriteride reduction of the 4-keto derivative.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
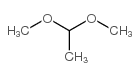 |
Dimethyl Acetal
CAS:534-15-6 |
C4H10O2 |
|
One-pot enol silane formation-Mukaiyama aldol-type addition ...
2008-04-18 [J. Org. Chem. 73(8) , 3299-302, (2008)] |
|
An expeditious synthesis of 4-hydroxy-2E-nonenal (4-HNE), it...
2007-12-01 [Chem. Phys. Lipids 150(2) , 239-43, (2007)] |
|
Matrix isolation infrared and ab initio study of the conform...
2003-05-01 [Spectrochim. Acta. A. Mol. Biomol. Spectrosc. 59(7) , 1497-507, (2003)] |
|
Hybrid Lithium-Sulfur Batteries with a Solid Electrolyte Mem...
2015-08-05 [ACS Appl. Mater. Interfaces 7 , 16625-31, (2015)] |
|
Mice lacking mitochondrial ferritin are more sensitive to do...
2014-08-01 [J. Mol. Med. 92(8) , 859-69, (2014)] |