Synthetic studies on perophoramidine and the communesins: construction of the vicinal quaternary stereocenters.
Jae Hong Seo, Gerald D Artman, Steven M Weinreb
Index: J. Org. Chem. 71(23) , 8891-900, (2006)
Full Text: HTML
Abstract
An efficient synthetic strategy for installation of the two vicinal quaternary carbon centers of the communesins is reported. Key steps include the O-allylation/Claisen rearrangement of spirolactone systems, which are formed by tandem intramolecular Heck cyclization/carbonylation. Substituent and solvent effects on the stereochemical outcome of the Claisen rearrangements have been examined. The stereochemical assignment of the allyl spirolactone previously reported as 17 has now been revised to 31, which has the communesin relative configuration at the quaternary carbons. Key C-allyl spirolactone 59 bearing functional handles required for the communesin core has been constructed with a 9.8:1 diastereomer ratio.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
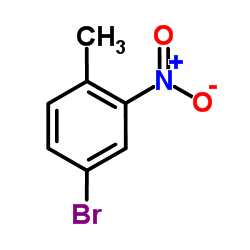 |
4-Bromo-2-nitrotoluene
CAS:60956-26-5 |
C7H6BrNO2 |
|
Synthesis of (+/-)-eusynstyelamide A.
2010-06-04 [Org. Lett. 12(11) , 2664-7, (2010)] |
|
Synthesis of 4-Bromo-2-chlorotoluene. Xue XM, et al.
[Chin. J. Pharm. 37(9) , 588, (2006)] |
|
A Simple, Safe and Efficient Synthesis of Tyrian Purple (6, ...
[Molecules 15(8) , 5561-5580, (2010)] |
|
Tyrian purple: 6, 6'-dibromoindigo and related compounds. Co...
[Molecules 6(9) , 736-769, (2001)] |
|
The bromo-2-nitrobenzoic acids. Erickson JLE, et al.
[J. Am. Chem. Soc. 74(22) , 5621-5623, (1952)] |