Synthesis and biological evaluation of new 3-(6-hydroxyindol-2-yl)-5-(Phenyl) pyridine or pyrazine V-Shaped molecules as kinase inhibitors and cytotoxic agents.
Pamela Kassis, Joanna Brzeszcz, Valérie Bénéteau, Olivier Lozach, Laurent Meijer, Rémi Le Guével, Christiane Guillouzo, Krzysztof Lewiński, Stéphane Bourg, Lionel Colliandre, Sylvain Routier, Jean-Yves Mérour
Index: Eur. J. Med. Chem. 11th ed., 46 , 5416-5434, (2011)
Full Text: HTML
Abstract
We here report the synthesis and biological evaluation of new 3-[(2-indolyl)]-5-phenyl-3,5-pyridine, 3-[(2-indolyl)]-5-phenyl-2,4-pyridine and 3-[(2-indolyl)]-5-phenyl-2,6-pyrazine derivatives designed as potential CDK inhibitors. Indoles and phenyls were used to generate several substitutions of the pyridine and pyrazine rings. The synthesis included Stille or Suzuki type reactions, which were carried out on the 3,5-dibromopyridine, 2,4-dichloropyridine and 2,6-dichloro-1-4-pyrazine moieties. Cell effects of the V-shaped family were in the micromolar range. Kinase assays were conducted and showed that compound 11 inhibited CDK5 with an inhibitory concentration of 160 nM with a moderate selectivity over GSK3 compared to the reference C which exhibited a slightly lower activity on CDK5 (1.5 μM). Compound 11 was also found to be the most potent compound in the series and was identified as a new lead for DYRK1A inhibitor discovery (IC(50) = 60 nM). Docking studies were carried out in order to investigate the inhibition of DYRK1A.Copyright © 2011 Elsevier Masson SAS. All rights reserved.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
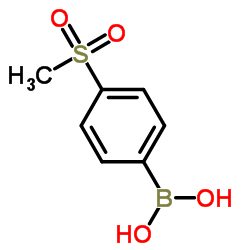 |
4-(methylsulfonyl)phenylboronic acid
CAS:149104-88-1 |
C7H9BO4S |
|
3,5-Diaryl-2-aminopyridines as a novel class of orally activ...
2012-04-12 [J. Med. Chem. 7th ed., 55 , 3479-3487, (2012)] |
|
Copper-catalyzed oxidative trifluoromethylthiolation of aryl...
2012-03-05 [Angew. Chem. Int. Ed. Engl. 10th ed., 51 , 2492-2495, (2012)] |
|
Synthesis of unsymmetrical 3,4-diaryl-3-pyrrolin-2-ones util...
2011-10-21 [J. Org. Chem. 76 , 8203-8214, (2011)] |
|
Highly selective rhodium-catalyzed conjugate addition reacti...
2007-11-09 [J. Org. Chem. 72 , 8870, (2007)] |
|
The design, synthesis and biological evaluations of C-6 or C...
2012-01-01 [Bioorg. Med. Chem. 1st ed., 20 , 467-479, (2012)] |