| Structure | Name/CAS No. | Articles |
|---|---|---|
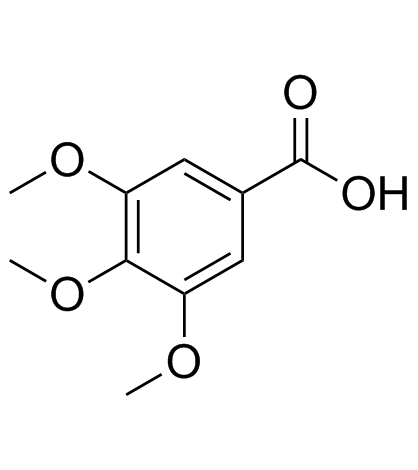 |
Trimethylgallic acid
CAS:118-41-2 |
|
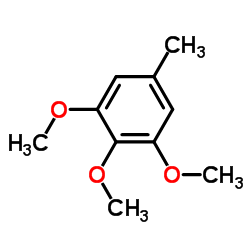 |
1,2,3-Trimethoxy-5-methylbenzene
CAS:6443-69-2 |