| Structure | Name/CAS No. | Articles |
|---|---|---|
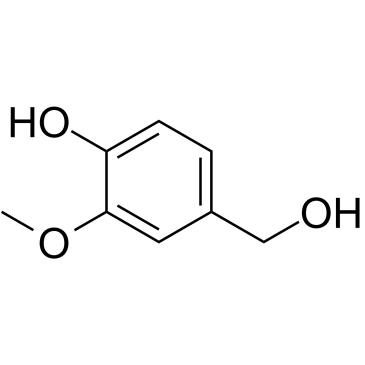 |
Vanillyl alcohol
CAS:498-00-0 |
|
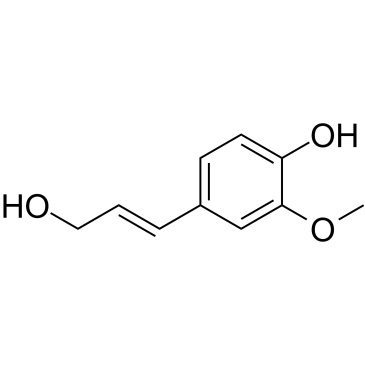 |
Coniferyl alcohol
CAS:458-35-5 |
|
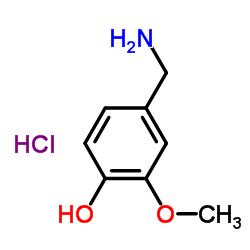 |
4-Hydroxy-3-methoxybenzylamine hydrochloride
CAS:7149-10-2 |