| Structure | Name/CAS No. | Articles |
|---|---|---|
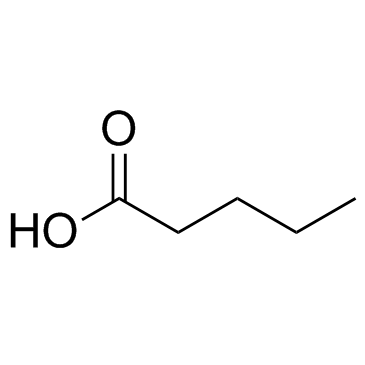 |
Pentanoic acid
CAS:109-52-4 |
|
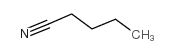 |
Valeronitrile
CAS:110-59-8 |