| Structure | Name/CAS No. | Articles |
|---|---|---|
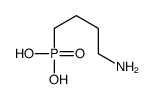 |
(4-Aminobutyl)phosphonic acid
CAS:35622-27-6 |
|
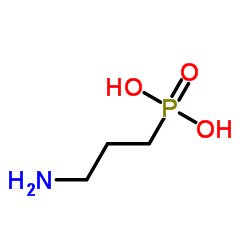 |
3-Aminopropylphosphonic acid
CAS:13138-33-5 |