A concerted structural transition in the plasminogen activator inhibitor-1 mechanism of inhibition.
Grant E Blouse, Michel J Perron, Jannah H Thompson, Duane E Day, Chad A Link, Joseph D Shore
Index: Biochemistry 41(40) , 11997-2009, (2002)
Full Text: HTML
Abstract
The inhibition mechanism of serpins requires a change in structure to entrap the target proteinase as a stable acyl-enzyme complex. Although it has generally been assumed that reactive center loop insertion and associated conformational change proceeds in a concerted manner, this has not been demonstrated directly. Through the substitution of tryptophan with 7-azatryptophan and an analysis of transient reaction kinetics, we have described the formation of an inhibited serpin-proteinase complex as a single concerted transition of the serpin structure. Replacement of the four tryptophans of plasminogen activator inhibitor type-1 (PAI-1) with the spectrally unique analogue 7-azatryptophan permitted observations of conformational changes in the serpin but not those of the proteinase. Formation of covalent acyl-enzyme complexes, but not noncovalent Michaelis complexes, with tissue-type plasminogen activator (t-PA) or urokinase (u-PA) resulted in rapid decreases of fluorescence coinciding with insertion of the reactive center loop and expansion of beta-sheet A. Insertion of an octapeptide consisting of the P14-P7 residues of the reactive center loop into beta-sheet A produced the same conformational change in serpin structure measured by 7-azatryptophan fluorescence, suggesting that introduction of the proximal loop residues induces the structural rearrangement of the serpin molecule. The atom specific modification of the tryptophan indole rings through analogue substitution produced a proteinase specific effect on function. The reduced inhibitory activity of PAI-1 against t-PA but not u-PA suggested that the mechanism of loop insertion is sensitive to the intramolecular interactions of one or more tryptophan residues.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
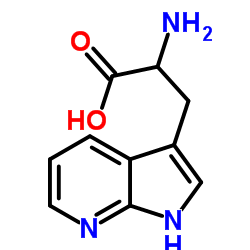 |
7-azatryptophan
CAS:7303-50-6 |
C10H11N3O2 |
|
Phosphorescence and optically detected magnetic resonance ch...
1998-06-23 [Biochemistry 37(25) , 8954-64, (1998)] |
|
Blue fluorescent amino acids as in vivo building blocks for ...
2010-02-15 [ChemBioChem. 11(3) , 305-14, (2010)] |
|
In vivo properties of thiol inhibitors of the three vasopept...
2004-02-01 [J. Pept. Res. 63(2) , 99-107, (2004)] |
|
Biosynthetic incorporation of tryptophan analogues into stap...
1997-03-01 [Protein Sci. 6(3) , 689-97, (1997)] |
|
Incorporation of the fluorescent amino acid 7-azatryptophan ...
2004-06-01 [Protein Sci. 13(6) , 1489-502, (2004)] |