Blue fluorescent amino acids as in vivo building blocks for proteins.
Lars Merkel, Michael G Hoesl, Marcel Albrecht, Andreas Schmidt, Nediljko Budisa
Index: ChemBioChem. 11(3) , 305-14, (2010)
Full Text: HTML
Abstract
In vivo expression of colored proteins without post-translational modification or chemical functionalization is highly desired for protein studies and cell biology. Cell-permeable tryptophan analogues, such as azatryptophans, have proved to be almost ideal isosteric substitutes for natural tryptophan in cellular proteins. Their unique spectral features, such as markedly red-shifted fluorescence, are transmitted into protein structures upon incorporation. Among the azaindoles under study (2-, 4-, 5-, 6-, and 7-azaindole) 4-azaindole has exhibited the largest Stokes shift (approximately 130 nm) in steady-state fluorescence measurements. It is also highly biocompatible and as 4-azatryptophan it can be translated into target protein sequences. However, its quantum yield and fluorescence intensity are still significantly lower when compared with natural indole/tryptophan. Since azatryptophans are hydrophilic, their presence in the hydrophobic core of proteins could be harmful. In order to overcome these limitations we have performed nitrogen methylation of azaindoles and generated mono- and dimethylated azaindoles. Some of these methyl derivatives retain the pronounced red shift present in the parent 4-azaindole, but with much higher fluorescence intensity (reaching the level of indole/tryptophan). Therefore, the blue fluorescence of azaindole-containing proteins could be further enhanced by the use of methylated analogues. Further substitution of any azaindole ring with either endo- or exocyclic nitrogen will not yield a spectral fluorescence maximum shift beyond 450 nm under steady-state conditions in the physiological milieu. However, green fluorescence is a special feature of tautomeric species of azaindoles in various nonaqueous solvents. Thus, the design or evolution of the protein interior combined with the incorporation of these azaindoles might lead to the generation of specific chromophore microenvironments that facilitate tautomeric or protonated/deprotoned states associated with green fluorescence.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
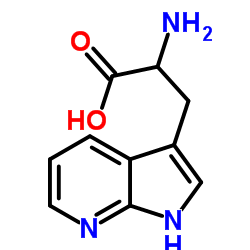 |
7-azatryptophan
CAS:7303-50-6 |
C10H11N3O2 |
|
Phosphorescence and optically detected magnetic resonance ch...
1998-06-23 [Biochemistry 37(25) , 8954-64, (1998)] |
|
In vivo properties of thiol inhibitors of the three vasopept...
2004-02-01 [J. Pept. Res. 63(2) , 99-107, (2004)] |
|
Biosynthetic incorporation of tryptophan analogues into stap...
1997-03-01 [Protein Sci. 6(3) , 689-97, (1997)] |
|
Incorporation of the fluorescent amino acid 7-azatryptophan ...
2004-06-01 [Protein Sci. 13(6) , 1489-502, (2004)] |
|
Influence of steric bulk and electrostatics on the hydroxyla...
1999-12-07 [Biochemistry 38(49) , 16283-9, (1999)] |