| Structure | Name/CAS No. | Articles |
|---|---|---|
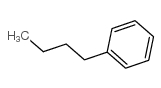 |
Butylbenzene
CAS:104-51-8 |
|
 |
octene
CAS:111-66-0 |