| Structure | Name/CAS No. | Articles |
|---|---|---|
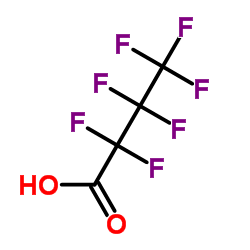 |
Heptafluorobutyric acid
CAS:375-22-4 |
|
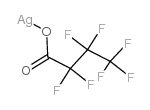 |
Butanoic acid,2,2,3,3,4,4,4-heptafluoro-, silver(1+) salt (1:1)
CAS:3794-64-7 |