| Structure | Name/CAS No. | Articles |
|---|---|---|
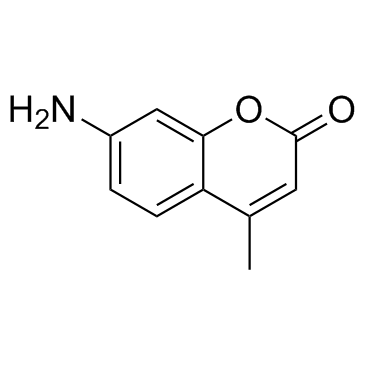 |
7-Amino-4-methylcoumarin
CAS:26093-31-2 |
|
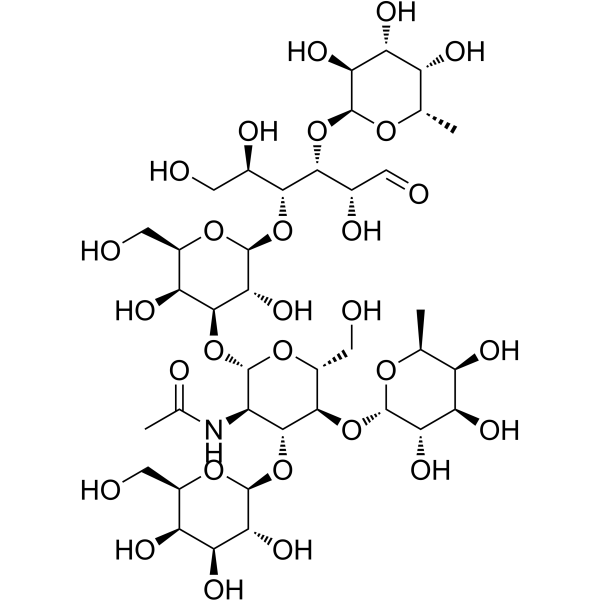 |
GAL-BETA1,3(FUC-ALPHA1,4)GLCNAC-BETA1,3GAL-BETA1,4(FUC-ALPHA1,3)GLC
CAS:62258-12-2 |