| Structure | Name/CAS No. | Articles |
|---|---|---|
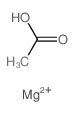 |
Magnesium acetate
CAS:142-72-3 |
|
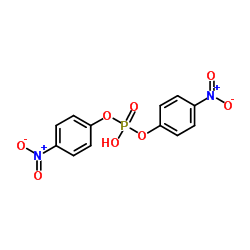 |
Bis(4-nitrophenyl) phosphate
CAS:645-15-8 |