Cyheptamide and 3-hydroxy-3-phenacyloxindole: structural similarity to diphenylhydantoin as the basis for anticonvulsant activity.
P W Codding, T A Lee, J F Richardson
Index: J. Med. Chem. 27(5) , 649-54, (1984)
Full Text: HTML
Abstract
The molecular structures of cyheptamide and 3-hydroxy-3- phenacyloxindole were determined by X-ray diffraction methods. The amide group in both compounds exhibits delocalization of the pi-electrons over the three atoms (N, C, and O), while the bond linking the amide to the tetrahedral carbon atom is a single bond. These structural features are also present in two drugs used for the treatment of generalized tonic-clonic ( GTC ) seizures, namely, carbamazepine and diphenylhydantoin. The shapes of cyheptamide , 3-hydroxy-3- phenacyloxindole , and carbamazepine have three features that are the same and can be simultaneously overlapped, the amide and two hydrophobic regions, whereas diphenylhydantoin fits two of the three regions at one time. These structural and electronic features are analyzed in light of current models for anticonvulsant activity.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
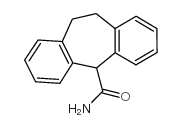 |
Cyheptamide
CAS:7199-29-3 |
C16H15NO |
|
A simple, efficient, and sensitive method for simultaneous d...
2015-11-01 [Antimicrob. Agents Chemother. 59 , 6682-8, (2015)] |
|
Evaluation of atazanavir and darunavir interactions with lip...
2014-08-01 [J. Pharm. Sci. 103(8) , 2520-9, (2014)] |
|
Anticonvulsant activities and brain concentrations of cyhept...
1980-01-01 [Proc. West. Pharmacol. Soc. 23 , 75-9, (1980)] |
|
Comparison of anticonvulsant potencies of cyheptamide, carba...
1981-06-01 [J. Pharm. Sci. 70(6) , 618-20, (1981)] |
|
A sensitive capillary GC-MS method for analysis of topiramat...
2012-09-01 [Biomed. Chromatogr. 26(9) , 1071-6, (2012)] |