A convergent Pd-catalyzed asymmetric allylic alkylation of dl- and meso-divinylethylene carbonate: enantioselective synthesis of (+)-australine hydrochloride and formal synthesis of isoaltholactone.
Barry M Trost, Aaron Aponick, Benjamin N Stanzl
Index: Chemistry 13(34) , 9547-60, (2007)
Full Text: HTML
Abstract
The use of a mixture of dl- and meso-divinylethylene carbonate as an electrophile in palladium-catalyzed asymmetric allylic alkylation reactions is reported. From the diastereomeric mixture of meso and chiral racemic starting materials, a single product is obtained in high optical purity employing either oxygen or nitrogen nucleophiles. The resulting dienes have proven to be versatile synthetic intermediates as each carbon is functionalized for further transformation and differentiated by virtue of the reaction. A mechanism for this intriguing transformation is proposed and a concise enantioselective total synthesis of (+)-australine hydrochloride is reported as well as a formal synthesis of isoaltholactone.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
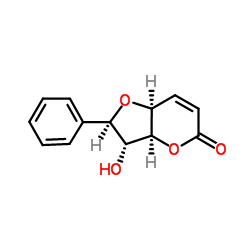 |
Altholactone
CAS:65408-91-5 |
C13H12O4 |
|
Stereoselective total synthesis of bioactive styryllactones ...
2008-01-04 [J. Org. Chem. 73(1) , 2-11, (2008)] |
|
The cytotoxicity of naturally occurring styryl lactones.
2006-02-01 [Phytomedicine 13(3) , 181-6, (2006)] |
|
Asymmetric synthesis of (+)-altholactone: a styryllactone is...
2008-01-01 [Chemistry 14(9) , 2842-9, (2008)] |
|
Stereospecificity in the Au-catalysed cyclisation of monoall...
2011-07-21 [Chem. Commun. (Camb.) 47(27) , 7659-61, (2011)] |
|
Altholactone induces apoptotic cell death in human colorecta...
2012-06-01 [Phytother Res. 26(6) , 926-31, (2012)] |