| Structure | Name/CAS No. | Articles |
|---|---|---|
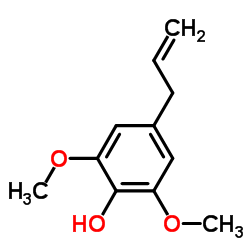 |
2,6-Dimethoxy-4-allylphenol
CAS:6627-88-9 |
|
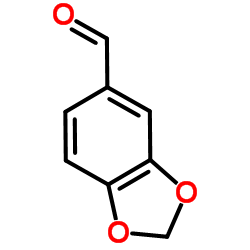 |
piperonal
CAS:120-57-0 |
|
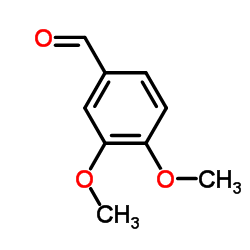 |
veratraldehyde
CAS:120-14-9 |