| Structure | Name/CAS No. | Articles |
|---|---|---|
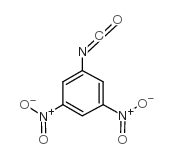 |
3,5-Dinitrophenyl isocyanate
CAS:59776-60-2 |
|
 |
(r)-(-)-1-(1-naphthyl)ethyl isocyanate
CAS:42340-98-7 |