Amination of tyrosine in liver cytosol protein of male F344 rats treated with 2-nitropropane, 2-nitrobutane, 3-nitropentane, or acetoxime.
R S Sodum, E S Fiala
Index: Chem. Res. Toxicol. 10(12) , 1420-6, (1997)
Full Text: HTML
Abstract
Previously, the secondary nitroalkane 2-nitropropane, a strong hepatocarcinogen in rats, had been shown to induce the formation of 8-aminoguanine in both DNA and RNA of rat liver through a sulfotransferase-mediated pathway. This pathway was postulated to convert the carcinogen into an aminating species [Sodum, R. S., et al. (1994) Chem. Res. Toxicol. 7, 344-351]. To submit this postulate to further test, we examined liver proteins of rats treated with 2-nitropropane, other carcinogenic secondary nitroalkanes, or the related rat liver tumorigen acetoxime for the presence of 3-aminotyrosine, the expected product of tyrosine amination. Using ion-pair and/or cation-exchange high-performance liquid chromatography with electrochemical detection, we found that the liver cytosolic proteins of these animals contained 0.1-1.5 mol of 3-aminotyrosine/10(3) mol of tyrosine. Treatment with the noncarcinogenic primary nitroalkane 1-nitropropane or with other primary nitroalkanes did not produce an analogous increase in the aminated amino acid (level of detection estimated at approximately 0.01 mol/10(3) mol of tyrosine). To our knowledge, this is the first report of the modification of protein tyrosine in vivo by a carcinogen. In vitro studies with acetoxime-O-sulfonate and hydroxylamine-O-sulfonate showed that these proposed intermediates in the activation pathway of 2-nitropropane react with guanosine to give 8-aminoguanosine, N1-aminoguanosine, and 8-oxoguanosine and also react with tyrosine to give 3-aminotyrosine and 3-hydroxytyrosine. The in vitro amination and oxidation of guanosine at C8 were also produced by acetophenoxime-O-sulfonate and 2-heptanoxime-O-sulfonate. These results provide additional evidence for the production of a reactive species capable of aminating nucleic acids and proteins from 2-nitropropane and other carcinogenic secondary nitroalkanes by a pathway involving oxime- and hydroxylamine-O-sulfonates as intermediates.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
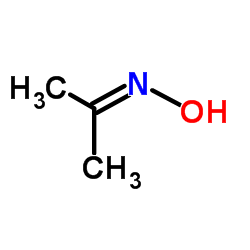 |
Propan-2-one oxime
CAS:127-06-0 |
C3H7NO |
|
Slow oxidation of acetoxime and methylethyl ketoxime to the ...
1999-02-01 [Toxicol. Sci. 47(2) , 144-50, (1999)] |
|
Synthesis and anticancer evaluation of benzyloxyurea derivat...
2014-01-01 [Chem. Pharm. Bull. 62(9) , 898-905, (2014)] |
|
Reversion of structure-activity relationships of antitumor p...
2007-01-01 [Mol. Pharmacol. 71(1) , 357-65, (2007)] |
|
Sex and organ differences in oxidative DNA and RNA damage du...
1990-09-01 [Carcinogenesis 11(9) , 1659-62, (1990)] |
|
Oxidative DNA and RNA damage in rat liver due to acetoxime: ...
1990-06-01 [Carcinogenesis 11(6) , 1013-6, (1990)] |