| Structure | Name/CAS No. | Articles |
|---|---|---|
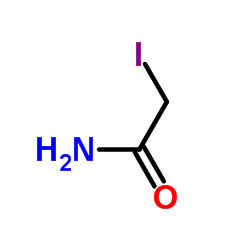 |
Iodoacetamide
CAS:144-48-9 |
|
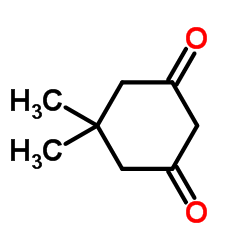 |
Dimedone
CAS:126-81-8 |
|
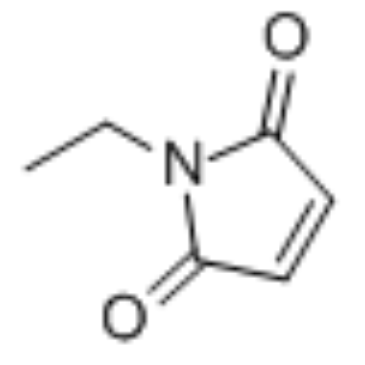 |
N-ethylmaleimide
CAS:128-53-0 |
|
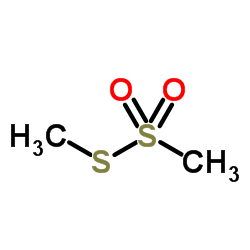 |
Methyl Methanethiosulfonate
CAS:2949-92-0 |
|
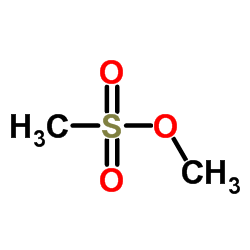 |
Methyl methanesulfonate
CAS:66-27-3 |