Elastase from activated human neutrophils activates procarboxypeptidase R.
Takeshi Kawamura, Noriko Okada, Hidechika Okada
Index: Microbiol. Immunol. 46(3) , 225-30, (2002)
Full Text: HTML
Abstract
Carboxypeptidase R (EC 3.4.17.20; CPR) is an unstable basic carboxypeptidase found in fresh serum in addition to carboxypeptidase N (CPN) which is a stable enzyme. CPR in fresh serum is generated from its zymogen (proCPR) during coagulation by trypsin-like enzymes such as thrombin and thrombin/thrombomodulin complexes. Since removal of the C-terminal arginine abrogates the anaphylatoxin activity of C3a and C5a, CPR and CPN are regarded as anaphylatoxin inactivators. We report here that the culture supernatant of activated human neutrophils converts proCPR to CPR. Addition of an elastase specific inhibitor, N-methoxysuccinyl-Ala-Ala-Pro-Val-chloromethyl ketone (MSAAPVCK) to the supernatant of stimulated neutrophils completely inhibited activation of proCPR. On the other hand, a thrombin specific inhibitor, p-Nitrophenyl-p'-amidinophenyl-methanesulfonate hydrochloride (pNP-pAPMS) inhibited only 16% of proCPR activation by the neutrophil supernatant. Furthermore, purified elastase converted proCPR to CPR. Therefore, elastase can activate proCPR directly, or indirectly through activation of some proteases, which have been contaminating in reagents. Release of CPR generating enzymes from neutrophils should play an important role in regulation of excess inflammation.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
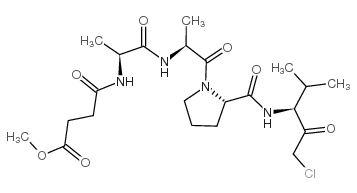 |
SPCK
CAS:65144-34-5 |
C22H35ClN4O7 |
|
Comparison of the effects of methoxysuccinyl-Ala-Ala-Pro-Val...
1992-12-01 [APMIS 100(12) , 1073-80, (1992)] |
|
Polymorphonuclear cells isolated from human peripheral blood...
1997-04-25 [J. Biol. Chem. 272(17) , 11079-87, (1997)] |
|
In vitro proteolysis of human plasma low density lipoprotein...
1986-02-15 [J. Biol. Chem. 261(5) , 2057-63, (1986)] |
|
Colistin stimulates the activity of neutrophil elastase and ...
2002-06-01 [Eur. Respir. J. 19(6) , 1136-41, (2002)] |
|
Proteolysis of gelatin-bound fibronectin by activated leukoc...
1991-10-01 [J. Leukoc. Biol. 50(4) , 331-40, (1991)] |