PR55 alpha, a regulatory subunit of PP2A, specifically regulates PP2A-mediated beta-catenin dephosphorylation.
Wen Zhang, Jun Yang, Yajuan Liu, Xi Chen, Tianxin Yu, Jianhang Jia, Chunming Liu
Index: J. Biol. Chem. 284 , 22649 - 22656, (2009)
Full Text: HTML
Abstract
A central question in Wnt signaling is the regulation of beta-catenin phosphorylation and degradation. Multiple kinases, including CKI alpha and GSK3, are involved in beta-catenin phosphorylation. Protein phosphatases such as PP2A and PP1 have been implicated in the regulation of beta-catenin. However, which phosphatase dephosphorylates beta-catenin in vivo and how the specificity of beta-catenin dephosphorylation is regulated are not clear. In this study, we show that PP2A regulates beta-catenin phosphorylation and degradation in vivo. We demonstrate that PP2A is required for Wnt/beta-catenin signaling in Drosophila. Moreover, we have identified PR55 alpha as the regulatory subunit of PP2A that controls beta-catenin phosphorylation and degradation. PR55 alpha, but not the catalytic subunit, PP2Ac, directly interacts with beta-catenin. RNA interference knockdown of PR55 alpha elevates beta-catenin phosphorylation and decreases Wnt signaling, whereas overexpressing PR55 alpha enhances Wnt signaling. Taken together, our results suggest that PR55 alpha specifically regulates PP2A-mediated beta-catenin dephosphorylation and plays an essential role in Wnt signaling.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
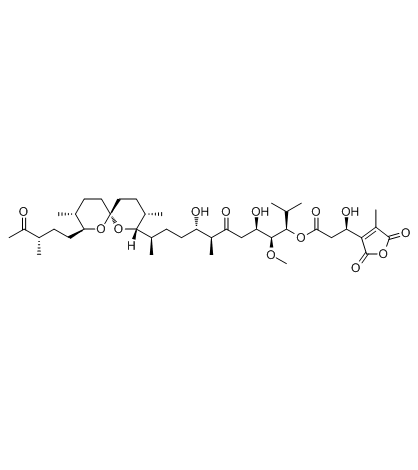 |
Tautomycin
CAS:109946-35-2 |
C41H66O13 |
|
Characterization of the tautomycetin biosynthetic gene clust...
2009-03-27 [J. Nat. Prod. 72(3) , 450-9, (2009)] |
|
The bifunctional glyceryl transferase/phosphatase OzmB belon...
2006-08-16 [J. Am. Chem. Soc. 128(32) , 10386-7, (2006)] |
|
Tautomycin suppresses growth and neuroendocrine hormone mark...
2009-01-01 [Am. J. Surg. 197(3) , 313-9, (2009)] |
|
Tautomycin's interactions with protein phosphatase 1.
2010-03-01 [Chem. Asian J. 5(3) , 410-20, (2010)] |
|
Functional characterization of ttmM unveils new tautomycin a...
2009-04-02 [Org. Lett. 11(7) , 1639-42, (2009)] |