| Structure | Name/CAS No. | Articles |
|---|---|---|
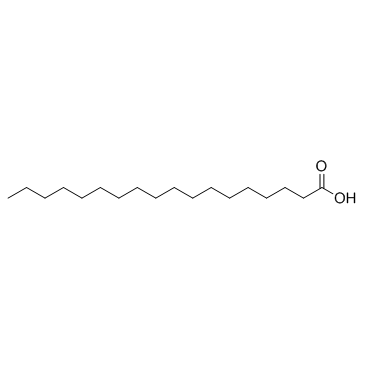 |
stearic acid
CAS:57-11-4 |
|
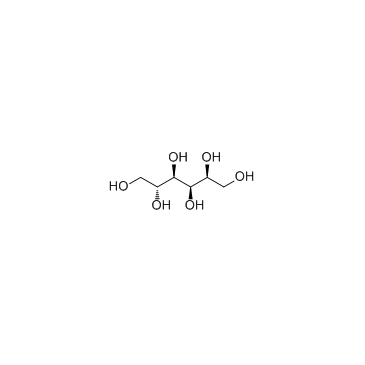 |
Sorbitol
CAS:50-70-4 |
|
 |
Methanol
CAS:67-56-1 |
|
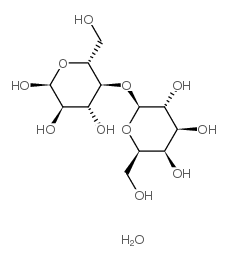 |
D-(+)-Lactose Monohydrate
CAS:64044-51-5 |
|
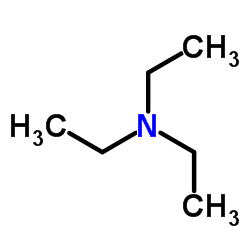 |
Triethylamine
CAS:121-44-8 |