| Structure | Name/CAS No. | Articles |
|---|---|---|
 |
sodium carbonate
CAS:497-19-8 |
|
 |
Copper sulfate pentahydrate
CAS:7758-99-8 |
|
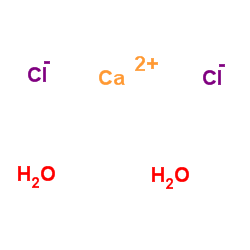 |
calcium chloride dihydrate
CAS:10035-04-8 |