|
~95% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~% |
|
~60% |

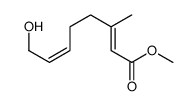
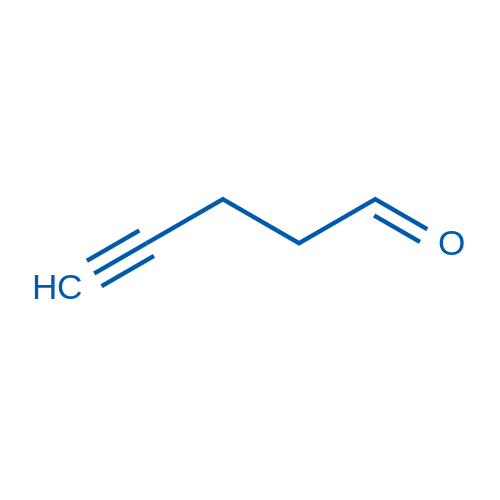
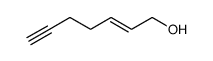
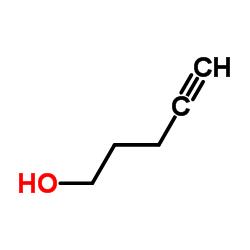
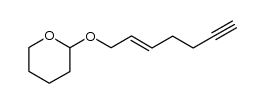

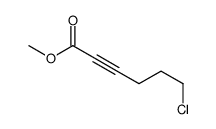


![2-Thiazolidinone,4-[(1R,4Z,8Z,10S,13R,15R)-15-hydroxy-5,10-dimethyl-3-oxo-2,14-dioxabicyclo[11.3.1]heptadeca-4,8-dien-15-yl]-,(4R) Structure](https://image.chemsrc.com/caspic/303/76343-94-7.png)