Mechanistic insight into cobalt-catalyzed stereodivergent semihydrogenation of alkynes: The story of selectivity control &z.star;
Xiaotian Qi, Xufang Liu, Ling-Bo Qu, Qiang Liu, Yu Lan
Index: 10.1016/j.jcat.2018.03.016
Full Text: HTML
Abstract
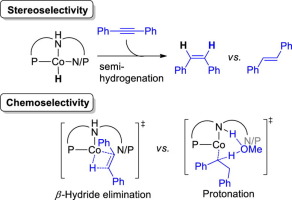
Selectivity control is a challenging and important subject in semihydrogenation of alkynes. Here, a combined theoretical and experimental study was performed to reveal the origin of the chemo- and stereoselectivity in cobalt-catalyzed stereodivergent semihydrogenation of alkynes. Three NNP and PNP type pincer ligands were considered in calculation. The computational results show that over-reduction of the alkene is forbidden in this catalytic system because the alkylcobalt(I) intermediate formed by alkene insertion prefers to undergo β-H elimination rather than protonation of the Co 
|
Combined quantitative FTIR and online GC study of Fischer-Tr...
2018-04-06 [10.1016/j.jcat.2018.03.026] |
|
Ethylene versus ethane: A DFT-based selectivity descriptor f...
2018-04-06 [10.1016/j.jcat.2018.03.019] |
|
Principles determining the activity of magnetic oxides for e...
2018-03-30 [10.1016/j.jcat.2018.03.012] |
|
Synthesis and characterization of Ag@Carbon core-shell spher...
2018-03-30 [10.1016/j.jcat.2018.02.029] |
|
Mechanistic investigations into the cyclopropanation of elec...
2018-03-30 [10.1016/j.jcat.2018.02.013] |