Preparation and characterization of the extracellular domain of human Sid-1.
Ashley J Pratt, Robert P Rambo, Pick-Wei Lau, Ian J MacRae
文献索引:PLoS ONE 7 , e33607, (2012)
全文:HTML全文
摘要
In C. elegans, the cell surface protein Sid-1 imports extracellular dsRNA into the cytosol of most non-neuronal cells, enabling systemic spread of RNA interference (RNAi) throughout the worm. Sid-1 homologs are found in many other animals, although for most a function for the protein has not yet been established. Sid-1 proteins are composed of an N-terminal extracellular domain (ECD) followed by 9-12 predicted transmembrane regions. We developed a baculovirus system to express and purify the ECD of the human Sid-1 protein SidT1. Recombinant SidT1 ECD is glycosylated and spontaneously assembles into a stable and discrete tetrameric structure. Electron microscopy (EM) and small angle x-ray scattering (SAXS) studies reveal that the SidT1 ECD tetramer is a compact, puck-shaped globular particle, which we hypothesize may control access of dsRNA to the transmembrane pore. These characterizations provide inroads towards understanding the mechanism of this unique RNA transport system from structural prospective.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
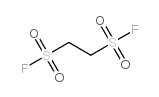 |
乙烯磺酰氟
CAS:677-25-8 |
C2H4F2O4S2 |
|
Electrochemical Fluorographane: Hybrid Electrocatalysis of B...
2015-11-09 [Chemistry 21 , 16474-8, (2015)] |
|
A simultaneous extraction method for organophosphate, pyreth...
2015-05-01 [Arch. Environ. Contam. Toxicol. 68(4) , 745-56, (2015)] |
|
Elastoviscous Transitions of Articular Cartilage Reveal a Me...
2015-01-01 [PLoS ONE 10 , e0143415, (2015)] |
|
Downregulation of caveolin‑1 upregulates the expression of g...
2016-01-01 [Mol. Med. Report. 13 , 744-52, (2015)] |
|
Phosphatidylethanolamine Is a Key Regulator of Membrane Flui...
2016-02-12 [J. Biol. Chem. 291 , 3658-67, (2016)] |