The study of controlling pore size on electrospun carbon nanofibers for hydrogen adsorption.
Ji Sun Im, Soo-Jin Park, Tae Jin Kim, Young Ho Kim, Young-Seak Lee
文献索引:J. Colloid. Interface Sci. 318(1) , 42-49, (2008)
全文:HTML全文
摘要
Polyacrylonitrile (PAN)-based carbon nanofibers (CNFs) were prepared by using electrospinning method and heat treatment to get the media for hydrogen adsorption storage. Potassium hydroxide and zinc chloride activations were conducted to increase specific surface area and pore volume of CNFs. To investigate the relation between pore structure and the capacity of hydrogen adsorption, textural properties of activated CNFs were studied with micropore size distribution, specific surface area, and total pore volume by using BET (Brunauer-Emmett-Teller) surface analyzer apparatus and the capacity of hydrogen adsorption was evaluated by PCT (pressure-composition-temperature) hydrogen adsorption analyzer apparatus with volumetric method. The surface morphology of activated CNFs was observed by SEM (scanning electron microscope) images to investigate the surface change through activation. Even though specific surface area and total pore volume were important factors for increasing the capacity of hydrogen adsorption, the pore volume which has pore width (0.6-0.7 nm) was a much more effective factor than specific surface area and pore volume in PAN-based electrospun activated CNFs.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
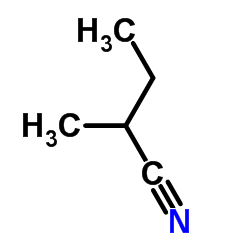 |
聚丙烯腈
CAS:25014-41-9 |
C5H9N |
|
Preparation and characteristics of electrospun activated car...
2007-10-01 [J. Colloid. Interface Sci. 314(1) , 32-37, (2007)] |
|
Prevention of dialysis-related amyloidosis using the polyacr...
1995-01-01 [Contrib. Nephrol. 112 , 137-44, (1995)] |
|
Superior prospect of chemically activated electrospun carbon...
[Mat. Res. Bul. 44(9) , 1871-1878, (2009)] |
|
The improved electrical conductivity of carbon nanofibers by...
[J. Ind. Eng. Chem. (Amsterdam, Neth.) 15(5) , 699-702, (2009)] |
|
Fluorination effects of carbon black additives for electrica...
[Carbon 47(11) , 2640-2647, (2009)] |