Conformational analysis of TOAC-labelled alamethicin F50/5 analogues.
Cristina Peggion, Micha Jost, Wim M De Borggraeve, Marco Crisma, Fernando Formaggio, Claudio Toniolo
文献索引:Chem. Biodivers. 4(6) , 1256-68, (2007)
全文:HTML全文
摘要
In the preceding paper in this issue, we reported the total syntheses in solution of a set of four TOAC-containing analogues of the [L-Glu(OMe)(7,18,19)] F50/5 component of alamethicin, the prototype of peptaibol antibiotics forming channels in the biological membranes. In this article, we have expanded this work to the examination of their preferred conformation in solution by use of a combination of CD, FT-IR absorption, and NMR spectroscopies. The results are strongly in favor of the view that replacement of the Aib residues at positions 1, 8, and 16 with TOAC (both are members of the helicogenic sub-class of C(alpha)-tetrasubstituted alpha-amino acids) does not significantly affect the overwhelmingly populated alpha-helical 3D structure of alamethicin. The X-ray diffraction crystal structure of the N(alpha)-protected, C-terminal, hexapeptide amide segment Z-L-Pro-L-Val-(Aib)(2)-[L-Glu(OMe)](2)-Fol lends further support to our conformational conclusions.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
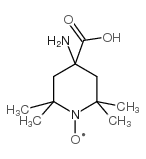 |
2,2,6,6-四甲基哌啶-1-氧基-4-氨基-4-羧酸
CAS:15871-57-5 |
C10H19N2O3 |
|
In silico interpretation of cw-ESR at 9 and 95 GHz of mono- ...
2011-11-10 [J. Phys. Chem. B 115(44) , 13026-36, (2011)] |
|
Intramembrane water associated with TOAC spin-labeled alamet...
2009-02-01 [Biophys. J. 96(3) , 997-1007, (2009)] |
|
Probing the helical tilt and dynamic properties of membrane-...
2012-03-01 [Biochim. Biophys. Acta 1818(3) , 645-50, (2012)] |
|
Membrane interaction of neuropeptide Y detected by EPR and N...
2005-08-15 [Biochim. Biophys. Acta 1714(2) , 103-13, (2005)] |
|
Electron paramagnetic resonance studies of an integral membr...
2006-09-20 [J. Am. Chem. Soc. 128(37) , 12070-1, (2006)] |