Identification, synthesis and quantification of process-related impurities in auraptene.
Yan-Gang Li, Hai-Fang Chen, Ming-Zhu Tu, Pu-Zhao Zhang, Xiao-Yun Wang, Jin-Bin Yuan, Wu-Liang Yang
文献索引:J. Pharm. Biomed. Anal. 56(2) , 191-9, (2011)
全文:HTML全文
摘要
Impurities in chemically synthesized auraptene, an active pharmaceutical ingredient (API), were detected by a gradient reverse-phase high-performance liquid chromatography (RP-HPLC) method. Molecular weights and major product ions of these chemical compounds were determined by liquid chromatography/Triple Quadrupole (LC-MS/MS) analysis. Structural assignments were presumed as umbelliferone (Imp-I), (E)-6,7-dihydroxy-3,7-dimethyl-2-octene-umbelliferone (Imp-II), (E)-6,7-epoxy-3,7-dimethyl-2-octene-umbelliferone (Imp-III) and 4-methylauraptene (Imp-IV). The impurities were authentically synthesized, confirmed by nuclear magnetic resonance spectroscopy (NMR) and infrared spectroscopy (IR), and subsequently used as reference samples in routing HPLC system suitability testing for method specificity and detectability. Method specificity was further verified by forced degradation studies. The developed method was validated for characterization of impurities in synthesized auraptene according to the guidelines of the International Conference on Harmonization (ICH) in our laboratory.Copyright © 2011 Elsevier B.V. All rights reserved.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
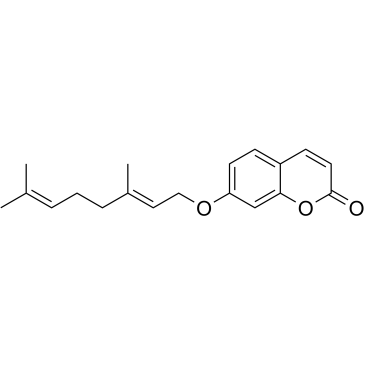 |
橙皮油内酯
CAS:495-02-3 |
C19H22O3 |
|
Suppression of mitochondrial respiration with auraptene inhi...
2015-11-10 [Oncotarget 6 , 38127-38, (2015)] |
|
Novel prodrugs for the treatment of colonic diseases based o...
2012-03-01 [Curr. Drug Deliv. 9(2) , 112-21, (2012)] |
|
Development of a quantitative bioassay to assess preventive ...
2011-08-01 [Nitric Oxide 25(2) , 183-94, (2011)] |
|
A natural propenoic acid derivative activates peroxisome pro...
2010-03-27 [Life Sci. 86(13-14) , 493-8, (2010)] |
|
Methyl galbanate, a novel inhibitor of nitric oxide producti...
2011-04-01 [J. Nat. Med. 65(2) , 353-9, (2011)] |