2-Alkenal-scavenging ability of m-diphenols.
Francisco J Hidalgo, Rosario Zamora
文献索引:Food Chem. 160 , 118-26, (2014)
全文:HTML全文
摘要
The reaction between m-diphenols (resorcinol, 2-methylresorcinol, 2,5-dimethylresorcinol, 3-methylphenol, orcinol, and phloroglucinol) and 2-alkenals (2-pentenal and 2-octenal) was studied in an attempt to understand the chemical pathways involved in the scavenging ability of m-diphenols for the 2-alkenals produced as a consequence of lipid oxidation. Phenols reacted chemically with 2-alkenals producing a number of 2H-chromenols, chromandiols, chromanols, and dihydropyrano[3,2-g]chromenes, which were isolated and identified by 1D and 2D nuclear magnetic resonance (NMR) spectroscopy and mass spectrometry (MS). The identification of all these compounds resulted in the construction of a general pathway for these reactions. These results confirm that the 2-alkenal-scavenging ability of m-diphenols is a consequence of their structure. This is a complex reaction in which many different products are formed. The most stable products were the chromandiols. However, the main reaction products were the 2H-chromenols. These products were unstable and disappeared as a consequence of polymerisation and browning reactions. Copyright © 2014 Elsevier Ltd. All rights reserved.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
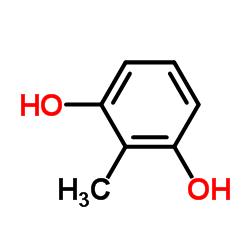 |
2,6-二羟基甲苯
CAS:608-25-3 |
C7H8O2 |
|
Cross-elicitation responses to 2-methoxymethyl-p-phenylenedi...
2015-04-01 [Br. J. Dermatol. 172(4) , 976-80, (2015)] |
|
Expanding cavitand chemistry: the preparation and characteri...
2001-04-17 [Chemistry 7(8) , 1637-45, (2001)] |
|
[Long-term preservation of DNA in aqueous solutions in the p...
2006-01-01 [Mikrobiologiia 75(5) , 662-9, (2006)] |
|
[Influence of chemical analogues of microbial autoregulators...
2006-01-01 [Mikrobiologiia 75(5) , 654-61, (2006)] |
|
A subchronic, teratologic, and dominant lethal study of 2-me...
1986-08-01 [Fundam. Appl. Toxicol. 7(2) , 293-8, (1986)] |