Kinetic studies on the oxidation of cytochrome b(5) Phe35 mutants with cytochrome c, plastocyanin and inorganic complexes.
Ping Yao, Yun-Hua Wang, Bing-Yun Sun, Yi Xie, Shun Hirota, Osamu Yamauchi, Zhong-Xian Huang
文献索引:J. Biol. Inorg. Chem. 7(4-5) , 375-83, (2002)
全文:HTML全文
摘要
To illustrate the functions of the aromatic residue Phe35 of cytochrome b(5) and to give further insight into the roles of the Phe35-containing hydrophobic patch and/or aromatic channel of cytochrome b(5), we studied electron transfer reactions of cytochrome b(5) and its Phe35Tyr and Phe35Leu variants with cytochrome c, with the wild-type and Tyr83Phe and Tyr83Leu variants of plastocyanin, and with the inorganic complexes [Fe(EDTA)](-), [Fe(CDTA)](-) and [Ru(NH(3))(6)](3+). The changes at Phe35 of cytochrome b(5) and Tyr83 of plastocyanin do not affect the second-order rate constants for the electron transfer reactions. These results show that the invariant aromatic residues and aromatic patch/channel are not essential for electron transfer in these systems.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
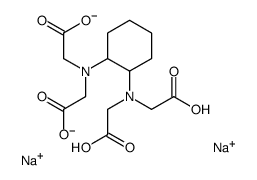 |
DCTA 钠盐
CAS:5786-78-7 |
C14H20N2Na2O8 |
|
Determination of synthetic ferric chelates used as fertilize...
2007-01-01 [J. Am. Soc. Mass Spectrom. 18(1) , 37-47, (2007)] |
|
Capillary electrophoretic-ultraviolet method for the separat...
2007-01-01 [J. AOAC Int. 90(3) , 834-7, (2007)] |
|
Detailed spectroscopic, thermodynamic, and kinetic studies o...
2009-08-17 [Inorg. Chem. 48(16) , 7864-84, (2009)] |
|
Direct magnetic resonance evidence for peroxymonocarbonate i...
2009-05-22 [J. Biol. Chem. 284(21) , 14618-27, (2009)] |
|
Improving sensitivity in simultaneous determination of coppe...
2006-03-31 [J. Chromatogr. A. 1110(1-2) , 261-7, (2006)] |