Common components of patch-clamp internal recording solutions can significantly affect protein kinase A activity.
G Vargas, T Y Yeh, D K Blumenthal, M T Lucero
文献索引:Brain Res. 828(1-2) , 169-73, (1999)
全文:HTML全文
摘要
Common components of whole-cell internal recording solutions were tested both in vitro and in patch-clamp experiments for their effects on the activity of cAMP-dependent protein kinase. Potassium fluoride (KF), 440 mM trimethylamine chloride and exclusion of bovine serum albumin (BSA) decreased the activity of the enzyme, while ethylene glycol-bis (beta-aminoethyl ether) N,N,N',N'-tetraacetic acid (EGTA) and the potassium salts of aspartate, gluconate, methylsulfate and monobasic phosphate increased its activity. Addition of KF to the internal solution produced a hyperpolarizing shift in the V1/2 of Ih channel activation, consistent with the KF-induced reduction of protein kinase A activity. Therefore, consideration of the composition of internal solutions is warranted when studying channel physiology by patch-clamp techniques.Copyright 1999 Elsevier Science B.V.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
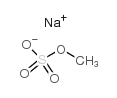 |
硫酸甲酯钠
CAS:512-42-5 |
CH3NaO4S |
|
Knoevenagel reaction in [MMIm][MSO₄]: synthesis of coumarins...
2011-01-01 [Molecules 16(6) , 4379-88, (2011)] |
|
Electronic structure of the sulfonyl and phosphonyl groups: ...
2007-10-15 [Inorg. Chem. 46(21) , 8871-86, (2007)] |
|
Engineering of choline oxidase from Arthrobacter nicotianae ...
2010-08-01 [Appl. Microbiol. Biotechnol. 87(5) , 1743-52, (2010)] |
|
Effects of inhibitors and substitutes for chloride in lumen ...
1999-03-01 [J. Pharmacol. Exp. Ther. 288(3) , 993-1001, (1999)] |
|
Acid-catalyzed dehydrative cyclization of 4-(D-galacto -pent...
2001-01-15 [Carbohydr. Res. 330(1) , 53-63, (2001)] |