Bovine renal cortex type I collagen: high contents of 3- and 4-hydroxyprolines.
S Fujiwara, Y Nagai
文献索引:J. Biochem. 89(5) , 1397-401, (1981)
全文:HTML全文
摘要
Type I collagen was prepared from bovine renal cortices by pepsin digestion followed by differential salt fractionation, and was identified by SDS-polyacrylamide gel electrophoresis, CM-cellulose chromatography, and by the analysis of CNBr-cleavage products of the alpha 1 chain. About 61-87% of total collagen in the tissue was solubilized by this procedure and type I collagen represents about 40% of the collagen solubilized. Renal cortex type I collagen is characteristic in that the extent of hydroxylation of the prolyl residues is high, but that of the lysyl residues is at the same level as in skin. Tissue-specific differences in the hydroxylation of prolyl residues of type I collagen are also discussed.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
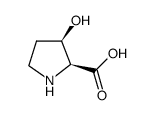 |
cis-3-hydroxy-L-proline
CAS:4298-05-9 |
C5H9NO3 |
|
Novel proline hydroxylase activities in the pneumocandin-pro...
2003-08-01 [Appl. Microbiol. Biotechnol. 62(2-3) , 263-7, (2003)] |
|
Insights on the evolution of prolyl 3-hydroxylation sites fr...
2011-01-01 [PLoS ONE 6(5) , e19336, (2011)] |
|
Conformational studies of peptides containing cis-3-hydroxy-...
2004-10-15 [J. Org. Chem. 69(21) , 7399-402, (2004)] |
|
Pneumocandins from Zalerion arboricola. V. Glutamic acid- an...
1992-12-01 [J. Antibiot. 45(12) , 1953-7, (1992)] |
|
Identification of 3-hydroxyproline residues in several prote...
1996-01-01 [Exp. Parasitol. 82(1) , 69-72, (1996)] |