Metal ion-binding studies highlight important differences between flaviviral RNA polymerases.
Isabelle Bougie, Martin Bisaillon
文献索引:Biochim. Biophys. Acta 1794(1) , 50-60, (2009)
全文:HTML全文
摘要
West Nile virus (WNV) is a member of the Flaviviridae family which includes a number of important human pathogens. The WNV NS5 protein harbors an RNA-dependent RNA polymerase activity which is required both for replication and transcription of the viral genome. To extend our studies on the role of metal ions in the activity of flaviviral polymerases, we have used fluorescence spectroscopy, circular dichroism, and a combination of chemical and thermal denaturation assays to monitor the consequences of metal ion binding to the enzyme. We demonstrate that the binding of magnesium is not critical for the structural stabilization of the enzyme. Moreover, structural studies indicate that the protein does not undergo conformational change upon the binding of magnesium ions. Additional binding assays also indicate that the interaction of magnesium ions with the enzyme does not significantly stimulate the interaction with the RNA or NTP substrates. The inability of cobalt hexamine, an exchange-inert metal complex structurally analogous to magnesium hexahydrate, to support the catalytic activity also allowed us to demonstrate a direct role of magnesium ions in the catalytic activity of the enzyme. Finally, a three-dimensional structural model of the active center of the enzyme was generated which highlighted the importance of two aspartate residues involved in the coordination of two metal ions. Mutational analyses confirmed the importance of these two amino acids for the binding of magnesium ions. Our data provide further insight into the precise role of magnesium ions for the RNA polymerase activity of the protein, and more importantly, highlight key differences between the RNA polymerases of the Flaviviridae family.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
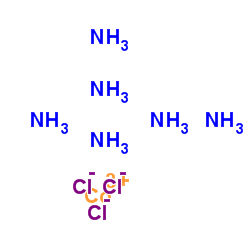 |
氯化六氨合钴
CAS:10534-89-1 |
H18Cl3CoN6 |
|
HCC ligation: rapid and specific DNA construction with blunt...
1986-12-22 [Nucleic Acids Res. 14(24) , 10118, (1986)] |
|
Iron regulatory element and internal loop/bulge structure fo...
1998-02-10 [Biochemistry 37(6) , 1505-12, (1998)] |
|
Protection of megabase DNA from shearing.
1995-10-11 [Nucleic Acids Res. 23(19) , 3999-4000, (1995)] |
|
The electron-transfer site of spinach plastocyanin.
1988-08-09 [Biochemistry 27(16) , 5876-84, (1988)] |
|
Reversible hypercondensation and decondensation of mitotic c...
2002-01-01 [J. Cell. Biochem. 85(2) , 422-34, (2002)] |