| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
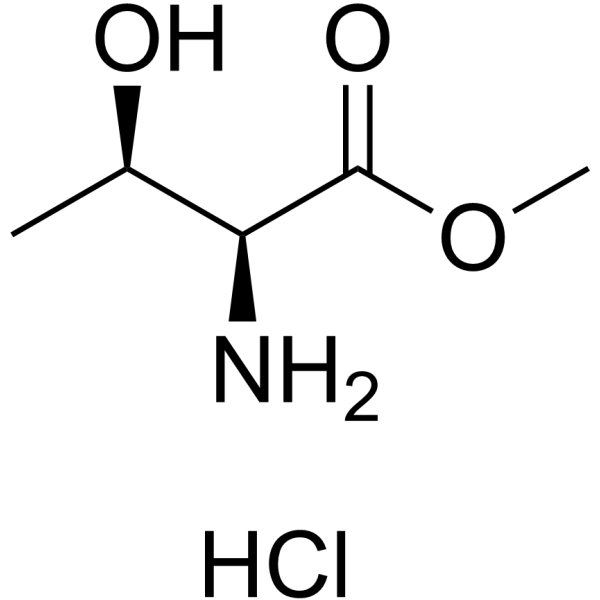 |
L-苏氨酸甲酯盐酸盐
CAS:39994-75-7 |
| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
 |
L-苏氨酸甲酯盐酸盐
CAS:39994-75-7 |