| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
 |
地喹碘铵
CAS:2019-42-3 |
|
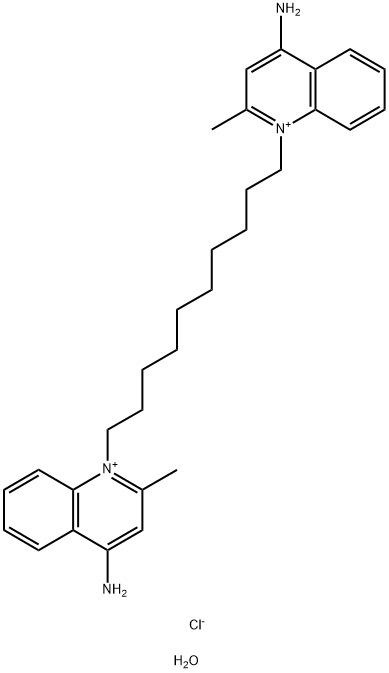 |
氯化异喹铵 水合物
CAS:1255077-34-9 |
|
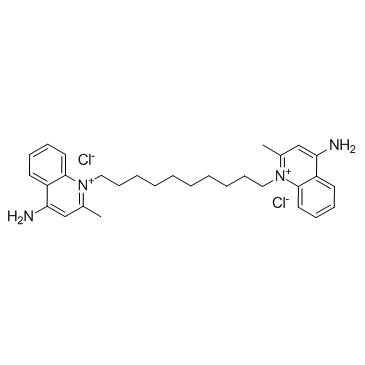 |
地喹氯铵
CAS:522-51-0 |
|
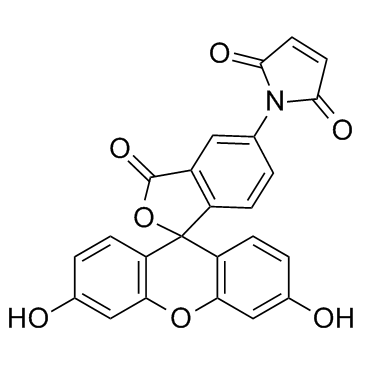 |
荧光素-5-马来酰亚胺
CAS:75350-46-8 |