| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
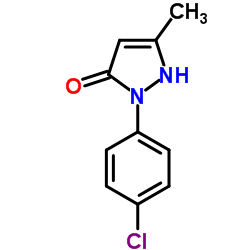 |
1-(4-氯基苯基)-3-甲基-5-吡唑酮
CAS:13024-90-3 |
| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
 |
1-(4-氯基苯基)-3-甲基-5-吡唑酮
CAS:13024-90-3 |