| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
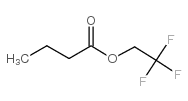 |
丁酸2,2,2-三氟乙酯
CAS:371-27-7 |
|
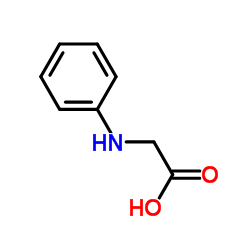 |
N-苯基甘氨酸
CAS:103-01-5 |
|
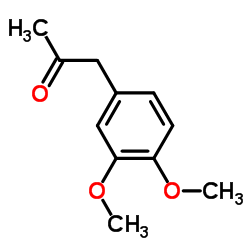 |
3,4-二甲氧基苯丙酮
CAS:776-99-8 |