| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
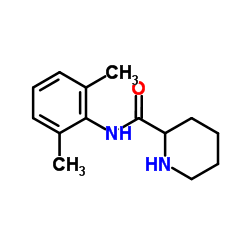 |
N-(2,6-二甲基苯基)哌啶-2-甲酰胺
CAS:15883-20-2 |
|
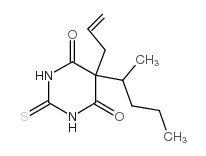 |
巴比妥
CAS:77-27-0 |