| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
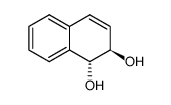 |
(1S,2S)-1,2-Dihydronaphthalene-1,2-diol
CAS:13011-97-7 |
|
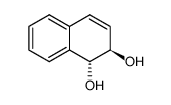 |
1,2-Naphthalenediol, 1,2-dihydro-, (1R,2R)-(-)
CAS:19216-89-8 |