Clinical pharmacokinetics of ketobemidone. Its bioavailability after rectal administration.
P Anderson, S Arnér, U Bondesson, L O Boréus, P Hartvig
文献索引:Eur. J. Clin. Pharmacol. 19(3) , 217-23, (1981)
全文:HTML全文
摘要
The pharmacokinetic constants and rectal bioavailability of the narcotic analgesic ketobemidone were determined in six male patients after surgery. Plasma concentrations were measured following intravenous administration of Ketogin 2 ml, containing ketobemidone chloride 10 mg, and a spasmolytic compound N,N-dimethyl-3,3-diphenyl-1-methylallylamine chloride 50 mg, and following rectal administration of one suppository of Ketogin, containing ketobemidone chloride 10 mg and the spasmolytic component 50 mg. Following intravenous administration, the disposition of ketobemidone followed a biexponential pattern with a fast distribution phase and a slower elimination phase: the plasma half-life (t1/2) was 2.42 +/- 0.41 h (m +/- SD). After rectal administration, the disposition of ketobemidone fitted a one-compartment model. The elimination half-life was 3.27 +/- 0.32 h. The mean rectal bioavailability for ketobemidone was 44% +/- 9%. The pharmacokinetic constants of the spasmolytic component, N,N-dimethyl-3,3-diphenyl-1-methylallylamine, were also determined in five of the patients, both after intravenous and after rectal administration. The plasma half-life was 3.07 +/- 0.53 h and 3.79 +/- 1.14 h, respectively. The rectal bioavailability was estimated to be 33% +/- 14%.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
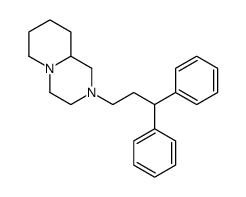 |
西酞普兰USP相关物质F
CAS:55011-89-7 |
C23H30N2 |
|
The mu1 and mu2 opioid receptor binding of ketobemidone, nor...
1996-08-01 [Pharmacol. Toxicol. 79(2) , 103-4, (1996)] |
|
Ketobemidone plus (RS)-3-dimethylamino-1,1-diphenylbut-1-ene...
1998-03-01 [Pharmacol. Toxicol. 82(3) , 157-60, (1998)] |
|
Analgesic effect and bioavailability of oral ketogan given a...
1988-01-01 [Acta Oncol. 27(5) , 583-9, (1988)] |
|
Premedication for day-case surgery: double-blind comparison ...
1986-10-01 [Acta Anaesthesiol. Scand. 30(7) , 502-6, (1986)] |
|
Histamine release from human basophils and isolated rat mast...
1982-01-01 [Acta Pharmacol. Toxicol. (Copenh.) 50(1) , 78-80, (1982)] |