A mechanism for hydroxylation by tyrosine hydroxylase based on partitioning of substituted phenylalanines.
P J Hillas, P F Fitzpatrick
文献索引:Biochemistry 35 , 6969, (1996)
全文:HTML全文
摘要
The iron-containing enzyme tyrosine hydroxylase catalyzes the hydroxylation of tyrosine to dihydroxyphenylalanine. A series of 4-X-substituted (X = H, F, Br, Cl, CH3, or CH3O) phenylalanines have been characterized as substrates to gain insight into the mechanism of hydroxylation. Multiple hydroxylated products were formed in most cases. As the size of the substituent at the 4-position increased, the site of hydroxylation switched from the 4- to the 3-position of the aromatic ring. The total amount of product formed with each amino acid showed a very good correlation with the sigma parameter of the substituent, with rho values of -4.3 +/- 0.7 or -5.6 +/- 0.8 when tetrahydrobiopterin or 6-methyltetrahydropterin, respectively, was used as cosubstrate. These values are consistent with a highly electron deficient transition state for hydroxylation. Oxygen addition at the 4-position resulted in either elimination of the substituent to form tyrosine or an NIH shift to form the respective 3-X-tyrosine. The relative amount of the product due to an NIH shift decreased in the order Br > CH3 > Cl >> F approximately CH3O approximately 0. A chemical mechanism for hydroxylation by tyrosine hydroxylase is presented to account for product formation from the various 4-substituted phenylalanines.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
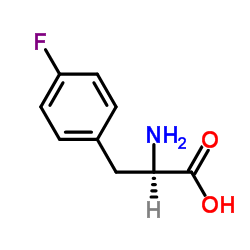 |
L-4-氟苯丙氨酸
CAS:1132-68-9 |
C9H10FNO2 |
|
Chemical genetics reveals a complex functional ground state ...
2007-05-01 [Nat. Chem. Biol. 3(5) , 268-273, (2007)] |
|
Genetic mapping of targets mediating differential chemical p...
2009-10-01 [Nat. Chem. Biol. 5 , 765-71, (2009)] |
|
Regulation of rat liver phenylalanine hydroxylase. I. Kineti...
1994-10-07 [J. Biol. Chem. 269 , 24637, (1994)] |
|
Chromosome condensing ability of mitotic proteins diminished...
1981-02-01 [Eur. J. Cell Biol. 23 , 312-316, (1981)] |