| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
 |
N-乙酰-D-蛋氨酸
CAS:1509-92-8 |
|
 |
N-乙酰-DL-蛋氨酸
CAS:1115-47-5 |
|
 |
N-乙酰-L-蛋氨酸
CAS:65-82-7 |
|
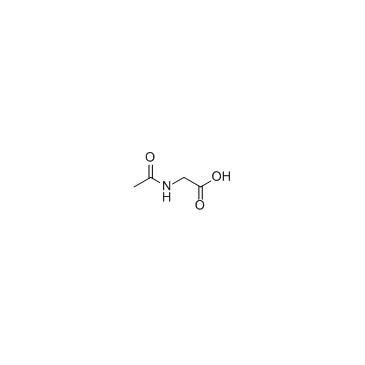 |
N-乙酰甘氨酸
CAS:543-24-8 |
|
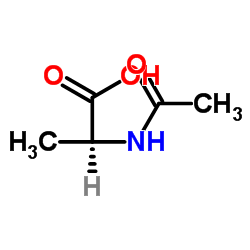 |
N-乙酰-L-丙氨酸
CAS:97-69-8 |