[Substrate specificity of T4 RNA-ligase: the role of a purine nucleotide base in forming a covalent AMP-RNA-ligase complex].
BA Iuodka, DIa Labe_kite, SI Sasnauskene
文献索引:Biokhimiia 58 , 857-865, (1993)
全文:HTML全文
摘要
The NTP binding site of bacteriophage T4 RNA-ligase (EC 6.5.1.3) was studied using several ATP analogs modified in the purine moiety of the nucleotide at positions 1, 2, 6 and 8: adenosine-N'-oxide 5'-triphosphate (I), 6-chloropurine riboside 5'-triphosphate (II), 6-mercaptopurine riboside 5'-triphosphate (III), 1,N6-ethenoadenosine 5'-triphosphate (IV), 8-sulphoadenosine 5'-triphosphate (V), 8-bromoadenosine 5'-triphosphate (VI), inosine 5'-triphosphate (VII) and guanosine 5'-triphosphate (VIII). For all of the ATP analogs under study a reversible inhibition was demonstrated. These analogs appeared to be competitive inhibitors of the T4 RNA-ligase adenylation reaction and only V, VI and VII were efficient as substrates. The kinetic parameters for the competitive inhibition, (I50, Ki) were determined. A putative structure for the T4 RNA-ligase active site was proposed.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
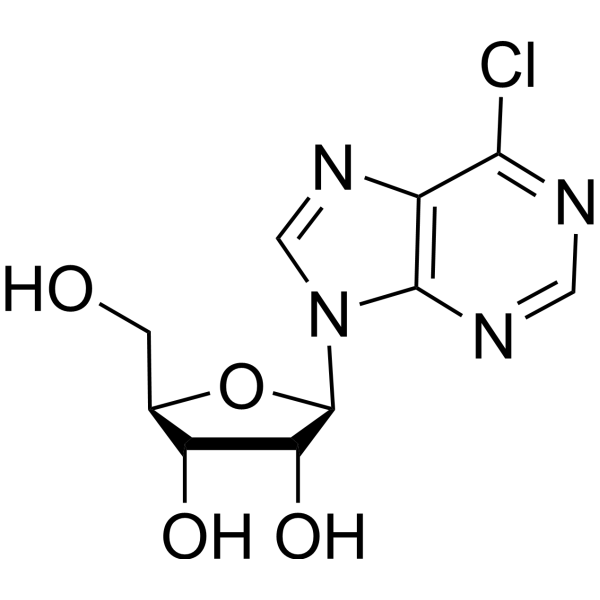 |
6-氯嘌呤核苷
CAS:5399-87-1 |
C10H11ClN4O4 |
|
Evidence on the existence of a purine ligand induced conform...
1982-08-31 [Biochemistry 21(18) , 4290-7, (1982)] |
|
Synthesis, biological evaluation and molecular modeling stud...
2007-05-15 [Biochem. Pharmacol. 73 , 1558-1572, (2007)] |
|
Synthesis and biological evaluation of nucleoside analogues ...
2007-05-01 [Bioorg. Med. Chem. Lett. 17 , 2470-3, (2007)] |
|
Introduction of a benzoyl group onto 6-chloropurine riboside...
2000-01-01 [Nucleic Acids Symp. Ser. 44 , 103-104, (2000)] |
|
Crystal structure of human type II inosine monophosphate deh...
1999-03-30 [Proc. Natl. Acad. Sci. U. S. A. 96 , 3531-3536, (1999)] |