| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
 |
4-硝基苯基-α-D-吡喃甘露糖苷
CAS:10357-27-4 |
|
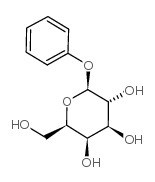 |
苯基β-D-吡喃半乳糖苷
CAS:2818-58-8 |