The influence of the peptide bond on the conformation of amino acids: a theoretical and FT-IR matrix-isolation study of N-acetylproline.
Bram Boeckx, Riet Ramaekers, Guido Maes
文献索引:Biophys. Chem. 159(2-3) , 247-56, (2011)
全文:HTML全文
摘要
A combined experimental matrix-isolation FT-IR and theoretical study has been performed to investigate the conformational behavior of N-acetylproline. The conformational landscape of N-acetylproline was explored using successively higher computational methods, i.e. HF, DFT(B3LYP) and finally MP2. The exploration resulted in 10 conformations with a relative energy difference smaller than 22 kJ.mol(-1) at the HF/3-21G level of theory. These conformations led to six different conformations after DFT(B3LYP) optimizations. Further optimization at the MP2/6-31++G** level of theory resulted in the same six conformations, all of them with an energy difference smaller than 11.5kJ.mol(-1). One conformation with an intramolecular H-bond was found which was energetically the most favorable conformation. The vibrational and thermodynamical features were calculated using the DFT and MP2 methodologies. In the experimental matrix-isolation FT-IR spectrum, the most stable conformation was dominant and at least two non-H-bonded conformations could be identified. An experimental rotamerization constant between the H-bonded and the other non-H-bonded conformations was estimated and appeared to agree reasonably well with the theoretical MP2 predictions. Some new spectral features of N-acetylproline compared to proline were discovered which might be used to discriminate between the acetylated and non-acetylated form.Copyright © 2011 Elsevier B.V. All rights reserved.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
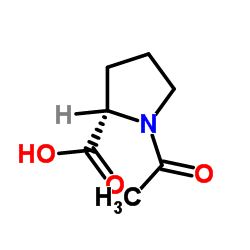 |
N-乙酰-L-脯氨酸
CAS:68-95-1 |
C7H11NO3 |
|
Quantum mechanical and NMR studies of ring puckering and cis...
2009-10-08 [J. Phys. Chem. A 113(40) , 10858-65, (2009)] |
|
Angiotensin-converting enzyme inhibitors. Mercaptan, carboxy...
1988-06-01 [J. Med. Chem. 31 , 1148-1160, (1988)] |
|
Synergistic binding of ligands to angiotensin-converting enz...
1988-03-25 [J. Biol. Chem. 263 , 4056-4068, (1988)] |
|
Conformational analysis of L-prolines in water.
2007-12-20 [J. Phys. Chem. B 111 , 14034-14042, (2007)] |
|
Purification and characterization of a novel aminoacylase fr...
2005-10-01 [Biosci. Biotechnol. Biochem. 69 , 1914-1922, (2005)] |